Keii, T. & Soga, K. E. d. Catalytic Olefin Polymerization Elsevier (2007).
Astruc, D. in Organometallic Chemistry and Catalysis Ch. 19, 395–408 (Springer, 2007).
Bertleff, W. inUllmann’s Encyclopedia of Industrial Chemistry Wiley-VCH Verlag GmbH & Co. KGaA (2000).
Van Leeuwen, P. W. N. M. & Claver, C. Rhodium Catalyzed Hydroformylation (Catalysis by Metal Complexes) Kluwer Academic Publishers (2000).
Diebolt, O., Müller, C. & Vogt, D. ‘On-water’ rhodium-catalysed hydroformylation for the production of linear alcohols. Catal. Sci. Technol. 2, 773–777 (2012).
Hebrard, F. & Kalck, P. Cobalt-catalyzed hydroformylation of alkenes: generation and recycling of the carbonyl species, and catalytic cycle. Chem. Rev. 109, 4272–4282 (2009).
Breit, B. Recent advances in alkene hydroformylation. Top Curr. Chem. 279, 139–172 (2007).
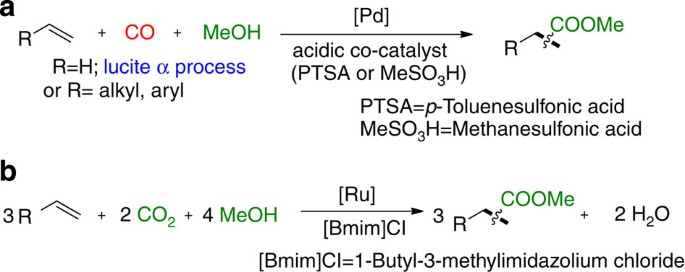
Kiss, G. Palladium-catalyzed reppe carbonylation. Chem. Rev. 101, 3435 (2001).
Jimenez Rodriguez, C., Foster, D. F., Eastham, G. R. & Cole-Hamilton, D. J. Highly selective formation of linear esters from terminal and internal alkenes catalysed by palladium complexes of bis-(di-tert-butylphosphinomethyl)benzene. Chem. Commun. 1720–1721 (2004).
Pugh, R. I., Drent, E. & Pringle, P. G. Tandem isomerisation-carbonylation catalysis: highly active palladium (II) catalysts for the selective methoxycarbonylation of internal alkenes to linear esters. Chem. Commun. 1476–1477 (2001).
Konrad, T. M., Fuentes, J. A., Slawin, A. M. Z. & Clarke, M. L. Highly enantioselective hydroxycarbonylation and alkoxycarbonylation of alkenes using dipalladium complexes as precatalysts. Angew. Chem. Int. Ed. 49, 9197–9200 (2010).
Morimoto, T. & Kakiuchi, K. Evolution of carbonylation catalysis: No need for carbon monoxide. Angew. Chem. Int. Ed. 43, 5580–5588 (2004).
Morimoto, T., Fuji, K., Tsutsumi, K. & Kakiuchi, K. CO-Transfer carbonylation reactions. a catalytic Pauson−Khand-Type reaction of enynes with aldehydes as a source of carbon monoxide. J. Am. Chem. Soc. 124, 3806–3807 (2002).
Park, J. H., Cho, Y. & Chung, Y. K. Rhodium-catalyzed Pauson–Khand-type reaction using alcohol as a source of carbon monoxide. Angew. Chem. Int. Ed. 49, 5138–5141 (2010).
Verendel, J. J., Nordlund, M. & Andersson, P. G. Selective metal-catalyzed transfer of H2 and CO from polyols to alkenes. Chem. Sus. Chem. 6, 426–429 (2013).
Konishi, H., Ueda, T., Muto, T. & Manabe, K. Remarkable improvement achieved by imidazole derivatives in ruthenium-catalyzed hydroesterification of alkenes using formates. Org. Lett. 14, 4722–4725 (2012).
Wieckowska, A., Fransson, R., Odell, L. R. & Larhed, M. Microwave-assisted synthesis of weinreb and MAP aryl amides via Pd-catalyzed heck aminocarbonylation using Mo(CO)6 or W(CO)6 . J. Org. Chem. 76, 978–981 (2011).
Sakakura, T., Choi, J.-C. & Yasuda, H. Transformation of carbon dioxide. Chem. Rev. 107, 2365–2387 (2007).
Aresta, M., Nobile, C. F., Albano, V. G., Forni, E. & Manassero, M. New nickel-carbon dioxide complex: synthesis, properties, and crystallographic characterization of (carbon dioxide)-bis(tricyclohexylphosphine)nickel. J. Chem. Soc. Chem. Commun. 636–637 (1975).
Mizuno, H., Takaya, J. & Iwasawa, N. Rhodium(I)-catalyzed direct carboxylation of arenes with CO2 via chelation-assisted C−H bond activation. J. Am. Chem. Soc. 133, 1251–1253 (2010).
Zhang, L., Cheng, J., Ohishi, T. & Hou, Z. Copper-catalyzed direct carboxylation of C-H bonds with carbon dioxide. Angew. Chem. Int. Ed. 49, 8670–8673 (2010).
Williams, C. M., Johnson, J. B. & Rovis, T. Nickel-catalyzed reductive carboxylation of styrenes using CO2 . J. Am. Chem. Soc. 130, 14936–14937 (2008).
Fujihara, T., Xu, T., Semba, K., Terao, J. & Tsuji, Y. Copper-catalyzed hydrocarboxylation of alkynes using carbon dioxide and hydrosilanes. Angew. Chem. Int. Ed. 50, 523–527 (2011).
Li, S., Yuan, W. & Ma, S. Highly regio- and stereoselective three-component nickel-catalyzed syn-hydrocarboxylation of alkynes with diethyl zinc and carbon dioxide. Angew. Chem. Int. Ed. 50, 2578–2582 (2011).
Fleischer, I. et al. From olefins to alcohols: efficient and regioselective ruthenium-catalyzed domino hydroformylation/reduction sequence. Angew. Chem. Int. Ed. 52, 2949–2953 (2013).
Takahashi, K., Yamashita, M., Tanaka, Y. & Nozaki, K. Ruthenium/C5Me5/bisphosphine- or bisphosphite-based catalysts for normal-selective hydroformylation. Angew. Chem. Int. Ed. 51, 4383–4387 (2012).
Wu, L., Fleischer, I., Jackstell, R. & Beller, M. Efficient and regioselective ruthenium-catalyzed hydroaminomethylation of olefins. J. Am. Chem. Soc. 135, 3989–3996 (2013).
Jenck, J., Kalck, P., Pinelli, E., Siani, M. & Thorez, A. Dinuclear ruthenium complexes as active catalyst precursors for the low pressure hydroformylation of alkenes into aldehydes. J. Chem. Soc. Chem. Commun. 1428–1430 (1988).
Clark, J. R., Griffiths, J. R. & Diver, S. T. Ruthenium hydride-promoted dienyl isomerization: access to highly substituted 1,3-dienes. J. Am. Chem. Soc. 135, 3327–3330 (2013).
Tominaga, K.-i. & Sasaki, Y. Ruthenium-catalyzed one-pot hydroformylation of alkenes using carbon dioxide as a reactant. J. Mol. Catal. A: Chem. 220, 159–165 (2004).
Srivastava, V. K. & Eilbracht, P. Ruthenium carbonyl-complex catalyzed hydroaminomethylation of olefins with carbon dioxide and amines. Catal. Commun. 10, 1791–1795 (2009).
Leung, J. C., Geary, L. M., Chen, T.-Y., Zbieg, J. R. & Krische, M. J. Direct, direct, redox-neutral prenylation and geranylation of secondary carbinol C–H bonds: C4-regioselectivity in ruthenium-catalyzed C–C couplings of dienes to α-hydroxy esters. J. Am. Chem. Soc. 134, 15700–15703 (2012).
Geary, L. M., Glasspoole, B. W., Kim, M. M. & Krische, M. J. Successive C–C coupling of dienes to vicinally dioxygenated hydrocarbons: ruthenium catalyzed [4+2] cycloaddition across the diol, hydroxycarbonyl, or dione oxidation levels. J. Am. Chem. Soc. 135, 3796–3799 (2013).
Leung, J. C. & Krische, M. J. Catalytic intermolecular hydroacylation of C–C p-bonds in the absence of chelation assistance. Chem. Sci. 3, 2202–2209 (2012).
Smejkal, T., Han, H., Breit, B. & Krische, M. J. All-carbon quaternary centers via ruthenium-catalyzed hydroxymethylation of 2-substituted butadienes mediated by formaldehyde: beyond hydroformylation. J. Am. Chem. Soc. 131, 10366–10367 (2009).
Srimani, D., Ben-David, Y. & Milstein, D. Direct synthesis of pyrroles by dehydrogenative coupling of β-aminoalcohols with secondary alcohols catalyzed by ruthenium pincer complexes. Angew. Chem. Int. Ed. 52, 4012–4015 (2013).
Dibenedetto, A., Stufano, P., Nocito, F. & Aresta, M. Ru(II)-Mediated hydrogen transfer from aqueous glycerol to CO2: from waste to value-added products. ChemSusChem 4, 1311–1315 (2011).
Behr, A., Kanne, U. & Keim, W. Alcohols as CO/H2 sources in ruthenium-catalyzed hydroesterification. J. Mol. Catal. 35, 19–28 (1986).
Olsen, E. P. K. & Madsen, R. Iridium-catalyzed dehydrogenative decarbonylation of primary alcohols with the liberation of syngas. Chem. Eur. J. 18, 16023–16029 (2012).
Gutel, T. et al. Influence of the self-organization of ionic liquids on the size of ruthenium nanoparticles: effect of the temperature and stirring. J. Mater. Chem. 17, 3290–3292 (2007).
Luska, K. L. & Moores, A. Ruthenium nanoparticle catalysts stabilized in phosphonium and imidazolium ionic liquids: dependence of catalyst stability and activity on the ionicity of the ionic liquid. Green Chem. 14, 1736–1742 (2012).
Lara, P., Philippot, K. & Chaudret, B. Organometallic ruthenium nanoparticles: a comparative study of the influence of the stabilizer on their characteristics and reactivity. Chem. Cat. Chem. 5, 28–45 (2013).